Some physical chemistry jokes collected by one of my p-chem students:
How many physical chemists does it take to wash a beaker?
None. That's what organic chemists are for!
A physical chemist is a student who goes to university thinking he might
want to be a physicist, but gets intimidated by the math.
Physical Chemistry is research on everything for which the negative
logaritm is linear with 1/T -- D.L. Bunker
Thanks to Alessandra!
On a historical note, D.L. Bunker is Don Bunker, a professor of physical chemistry at my alma mater, University of California, Irvine. He died suddenly during my freshman year.
- Home
- Angry by Choice
- Catalogue of Organisms
- Chinleana
- Doc Madhattan
- Games with Words
- Genomics, Medicine, and Pseudoscience
- History of Geology
- Moss Plants and More
- Pleiotropy
- Plektix
- RRResearch
- Skeptic Wonder
- The Culture of Chemistry
- The Curious Wavefunction
- The Phytophactor
- The View from a Microbiologist
- Variety of Life
Field of Science
-
-
Change of address10 months ago in Variety of Life
-
Change of address10 months ago in Catalogue of Organisms
-
-
Earth Day: Pogo and our responsibility1 year ago in Doc Madhattan
-
What I Read 20241 year ago in Angry by Choice
-
I've moved to Substack. Come join me there.1 year ago in Genomics, Medicine, and Pseudoscience
-
-
-
-
Histological Evidence of Trauma in Dicynodont Tusks7 years ago in Chinleana
-
Posted: July 21, 2018 at 03:03PM7 years ago in Field Notes
-
Why doesn't all the GTA get taken up?7 years ago in RRResearch
-
-
Harnessing innate immunity to cure HIV9 years ago in Rule of 6ix
-
-
-
-
-
-
post doc job opportunity on ribosome biochemistry!11 years ago in Protein Evolution and Other Musings
-
Blogging Microbes- Communicating Microbiology to Netizens11 years ago in Memoirs of a Defective Brain
-
Re-Blog: June Was 6th Warmest Globally11 years ago in The View from a Microbiologist
-
-
-
The Lure of the Obscure? Guest Post by Frank Stahl14 years ago in Sex, Genes & Evolution
-
-
Lab Rat Moving House14 years ago in Life of a Lab Rat
-
Goodbye FoS, thanks for all the laughs14 years ago in Disease Prone
-
-
Slideshow of NASA's Stardust-NExT Mission Comet Tempel 1 Flyby15 years ago in The Large Picture Blog
-
in The Biology Files
The Who, What, When, Where and Why of Chemistry
Chemistry is not a world unto itself. It is woven firmly into the fabric of the rest of the world, and various fields, from literature to archeology, thread their way through the chemist's text.
Cracking the Top 100 Podcasts
I found out today, quite by accident, that the podcast of my quantum mechanics lectures has been hovering in Apple iTunes Top 100 list! My students are not subscribing through iTunes (they are using other feed aggregators) - so is quantum chemistry suddenly fashionable?
Let's hope!
Let's hope!
When is red yellow?
 I promised my class that by the end of the semester they'd know why flamingos are pink and Cheetos are orange. In that vein, last week's quiz included a question asking them to figure out which of two labels belonged on a bottle of reddish violet solution and which went on the yellowish solution.
I promised my class that by the end of the semester they'd know why flamingos are pink and Cheetos are orange. In that vein, last week's quiz included a question asking them to figure out which of two labels belonged on a bottle of reddish violet solution and which went on the yellowish solution.One student correctly labled the bottles, but wondered if my choice of compounds was a red herring. She noted that the name of the compound that she predicted would produce the reddish solution (canthaxanthin) meant "yellow" in Greek. I told her that, in fact, I hadn't known that xanthin meant yellow and wasn't trying to mislead them!
This sent me on a hunt to discover why a reddish compound was named for "yellow". Turns out canthaxanthin is named for the mushroom species from which it was first extracted (Cantharellus cinnabarinus) and the Greek for yellow (xanthin). Cantharus is Latin for a two horned drinking cup, which the fungus resembles. The color can tend toward the orange, so that may be the source of the "yellow" in the name.
Weird Words of Science 7: nano dreams
Orac is dreaming of an iPod nano.
Where does nano come from? The root comes from the Greek for dwarf. The prefix was formally adopted in the late 1940s to mean 10-9, as in a nanometer. The construction had been floating around since the early part of the 20th century. The OED catalogs the first appearance in English in nanphanerophyte (a small shrub), but the French were using it about 50 years earlier.
Nano along now carries the connotation of the very small, typically on the nanometer scale. A bond between two carbon atoms is about 1/10 of a nanometer.
Where does nano come from? The root comes from the Greek for dwarf. The prefix was formally adopted in the late 1940s to mean 10-9, as in a nanometer. The construction had been floating around since the early part of the 20th century. The OED catalogs the first appearance in English in nanphanerophyte (a small shrub), but the French were using it about 50 years earlier.
Nano along now carries the connotation of the very small, typically on the nanometer scale. A bond between two carbon atoms is about 1/10 of a nanometer.
Quantum jokes?
Q: Why won't Heisenberg's operators live in the suburbs?
A: They don't commute.
To really understand the joke, it's helpful to know the general form of the Heisenberg uncertainty principle. The Robertson-Schroedinger inequality

leads directly to the more general statement of the Heisenberg uncertainty principle. The piece in the little square brackets [A,B] on the right hand side is called the commutator of the operators A and B. When A and B don't commute, [A,B] is non-zero and you end up with uncertain simultaneous measurements.
Commuting operators can be applied in any order and you get the same result.
My 9 year-old says that if you have to explain a joke, it's not a joke. And by this definition, he says, this post is not a joke!
A: They don't commute.
To really understand the joke, it's helpful to know the general form of the Heisenberg uncertainty principle. The Robertson-Schroedinger inequality

leads directly to the more general statement of the Heisenberg uncertainty principle. The piece in the little square brackets [A,B] on the right hand side is called the commutator of the operators A and B. When A and B don't commute, [A,B] is non-zero and you end up with uncertain simultaneous measurements.
Commuting operators can be applied in any order and you get the same result.
My 9 year-old says that if you have to explain a joke, it's not a joke. And by this definition, he says, this post is not a joke!
Flying Objects
One of the major uses of helium is to keep superconducting magnets very cold. The coils must be kept at a chilly 450 degrees Fahrenheit below zero in order to achieve the strong magnet fields necessary for magnetic resonance imaging. MRI is a widely used medical imaging technique, partly because the radiation used is of much lower energy than that need for CT scans, for example. MRI is actually an application of a quantum mechanical phenomena called NMR (nuclear magnetic resonance). The energy states of the nuclei in a magnetic field can be changed by tickling them with very small amounts of energy (radio waves to be precise), and the changes in these states can be used to produce an image of living tissue. The stronger the magnet field, the more easily detected the energy changes are, hence the use of the strong field superconducting magnets.
So how strong are these magnets? They are on the order of 1 Tesla, thousands of times more powerful than your basic horseshoe magnet. If you're not careful with a superconducting magnet of this strength, it can suck up a lot of metal. See the photos here for some stunning examples.
So how strong are these magnets? They are on the order of 1 Tesla, thousands of times more powerful than your basic horseshoe magnet. If you're not careful with a superconducting magnet of this strength, it can suck up a lot of metal. See the photos here for some stunning examples.
Strategic Reserves
President Bush recently announced that the strategic petroleum reserves would be breached to counter gasoline shortages in the wake of Hurricane Katrina. Did you know that government maintains strategic reserves of other materials as well? The strategic helium reserves are kept in Amarillo, TX in a natural formation called the Bush Dome. The He reserve was created in 1925, when the major strategic use was military dirigibles. Helium extraction is incidental to methane (natural gas) production. These days helium is used in magnetic resonance imaging to cool the coils in the magnets so they will superconduct and other cryogenic applications. The reserve contains about 30 billion scm (an scm is a cubic meter at standard conditions -- not a square cubic meter as one report states!)
Camille Minichino visited the chemistry department at Bryn Mawr this week. She is the author of the Periodic Table mystery series. In her second book, The Helium Murder, physicist Gloria Lamerino suspects Congresswoman Hurley has been murdered because of her position on the sale of helium in the reserves.
Standard conditions for a gas are 298 K and 1 atmosphere of pressure.
Camille Minichino visited the chemistry department at Bryn Mawr this week. She is the author of the Periodic Table mystery series. In her second book, The Helium Murder, physicist Gloria Lamerino suspects Congresswoman Hurley has been murdered because of her position on the sale of helium in the reserves.
Standard conditions for a gas are 298 K and 1 atmosphere of pressure.
The University Senate is not a Bath House

Mathematical Physics Seminar: Professor Hilbert, with the assistance of Dr E Noether, Mondays from 4-6, no tuition.
Winter 1916-17 catalog, University of Gottingen
Emmy Noether was one of the greatest mathematicians of the 20th century - indeed of any century. Born in 1882 in Erlangen, Germany, where her father was a professor of mathematics at the university, she is known for her work in ring theory and non-commutative algebras. Her work in the theory of invariants proved essential to Einstein in formulating his general theory of relativity. In the late 19th and early 20th century, women were not permitted to officially matriculate at German universities, though they could petition individual professors to attend their classes. Noether attended courses at Erlangen, then at Gottingen, receiving her Ph.D. from Gottingen in 1907. She returned to Erlangen to assist her father, but continued her own mathematical work (much of which was published in the papers of others). In 1915, Hilbert (yes, real analysis and quantum afficiandos, that Hilbert - of Hilbert space) persuaded her to return to Gottingen. Though Gottingen refused to appoint her officially to the faculty there until 1919, Noether taught courses by having them advertised under Hilbert's name. Hilbert argued strongly for Noether's addition to the faculty, famously proclaiming, "I do not see that the sex of the candidate is against her admission as a Privatdozent. After all, the university senate is not a bathhouse."
In 1933, she fled Nazi Germany, accepting a visiting position at Bryn Mawr College. She died at Bryn Mawr Hospital in 1935; her ashes are buried in the Cloisters at Bryn Mawr.
Quantum Chemistry in the Time of Cholera
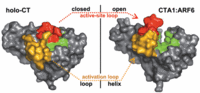
Slate magazine notes that occasional cases of cholera have been reported in the Gulf states, suggesting that the bacteria Vibrio cholerae is alive and well in that area. Recently, a group at Universtiy of Washington [O'Neal, Claire J., Jobling, Michael G., Holmes, Randall K., Hol, Wim G. J.
"Structural Basis for the Activation of Cholera Toxin by Human ARF6-GTP"
Science 2005 309: 1093-1096]has used x-ray crystallography to probe how the cholera toxin provokes such massive diarrhea when it takes up residence in the human gut. A figure from the paper shows how the overall 3-dimensional structure of the toxin changes when an activator protein binds to it. Protein chemists use the term "allosteric" to describe these mechanisms.
The picture is what interests me. I lectured on Monday about the probability density that you can compute using quantum mechanics (ψ*ψdτ). You can also measure the density experimentally using x-ray techniques, as Hol's group did for the cholera toxin. These sorts of pictures don't actually show the full density function, but a surface of constant electron density, usually around .002 e/bohr3, which seems small. A bohr is very small (0.529 x 10-10 )meters, so this corresponds to about one mole of electrons (on the order of 1023!) per cubic inch. This sounds like a lot, until you realize it's about the same as the number of electrons in a teaspoon of water!
That the Programmer may Return to being a Mathematician
The solution of differential equations has taken on an entirely new dimension in the 75+ years since Erwin Schrodinger proposed approaching the description of matter waves using PDEs. The solution of many differential equations can now be found numerically, using computers. Grace Hopper, who graduated with a BA in math from Vassar, a women's college, in 1928. She worked on her doctorate in math at Yale and returned to Vassar as a professor of mathematics in 1931. During WW II she joined the waves and was assigned to the team running the Mark I - an early digital computer. Her work there eventually led her to design the first compiler - a translator which turns the "natural" language of the programmer into the binary code that the computer can read. Hopper hoped that then "the programmer may return to being a mathematician." The development of widely used symbolic algebra programs such Mathematica and Maple can be traced to Grace Hopper's work. Legend has it that Grace Hopper is the one who coined the term "bug" for problems with a computer, after pulling a moth out of one of the machines.
Subscribe to:
Posts (Atom)















